Seventh Grade Science at TFAA
Welcome to the 2025–2026 school year! My name is Mr. Shelton, and I’m thrilled to be your 7th grade science teacher at Thurman Francis Arts Academy. I can’t wait to begin this journey with you—it’s truly an honor to be part of your educational experience.
A little about me: I’m married, a proud parent of three, and I love spending time outdoors. I’ve been in education for 26 years, serving as a classroom teacher, science program coordinator with Vanderbilt University, and assistant principal. Most recently, I spent five wonderful years teaching 7th grade science at Stewarts Creek Middle School.
My core belief is that open communication, trust, and mutual respect between students, parents, and teachers are essential for success. Please don’t hesitate to reach out with any questions or concerns—I’m always happy to connect and will respond as promptly as possible.
In my classroom, I emphasize positivity and motivation. I believe students thrive when they feel encouraged and valued. To that end, I use daily and weekly incentives like Jolly Ranchers for everyday achievements, popsicles for winning classroom games, and ice cream to celebrate consistent hard work and perseverance. These small rewards build excitement and encourage students to give their best effort.
I’m passionate about science and teaching, and I’m committed to making this year both fun and challenging. You can expect to be engaged in hands-on science practices, encouraged to think critically about the world around you, and inspired to look forward to class each day. I believe in making the most of every moment we have together—after all, there’s no substitute for time on task!
One phrase you’ll hear often in my class is: “If you know the song, you know the science!” Get ready to learn through music, collaborate with classmates, move around, explore strange phenomena, ask deep questions, build models, and dive into all that the 7th grade science curriculum has to offer.
Let’s go, Rams!
Sincerely,
Mr. Shelton
7th Grade Science Teacher
Thurman Francis Arts Academy
“IYKTSYKTS”





Matter and Interaction
1) Evaluate and communicate information that all substances in the universe are made of many
different types of atoms that combine in various ways.
2) Collect and analyze data about the physical properties of the components of a mixture to use as
evidence that the identities of the components change during a chemical reaction.
3) Develop a model to explain how changes to a system can be explained by changes in
temperature and/or pressure and the effect of those changes on particle motion and/or spatial
arrangement.
4) Use computational thinking to demonstrate that all atoms in the reactants are present in the
products of a chemical reaction supporting the Law of Conservation of Mass.
Types of Energy
1) Plan and carry out an investigation to demonstrate that the interaction between substances can
cause chemical reactions that release or store energy.
2) Develop a model to explain how food is utilized through chemical reactions to form new
molecules that support growth, resulting in the release of energy as matter moves through an
organism.
Molecules to Organisms
1) Develop models that identify and explain the structure and function of major cell organelles
and structures (i.e., vacuoles, chloroplasts, lysosomes, mitochondria, cell membrane, cell wall,
nucleus, cytoplasm) as they contribute to the life activities within a system.
2) Obtain information about the cellular structures of unicellular and multicellular organisms
across kingdoms and domains in order to compare how these structures support the functions
(i.e., obtain food, water, waste disposal, and the environment in which they live) of the
organism.
3) Develop and use a hierarchical model of a multicellular organism to explain that the body of
humans and other animals is a system of multiple interacting subsystems specialized for
56 particular body functions [e.g., digestion, respiration, excretion, circulation, sensation (nervous
and integumentary), locomotion (musculoskeletal), reproduction, and immunity].
4) Analyze data to determine the effect of genetic factors (e.g., specific breeds of organisms and
their typical sizes) and environmental factors (e.g., food and space availability) that influence the
growth of plants and animals.
5) Obtain and communicate information to provide evidence that illustrates the causal
relationships between information received by sensory receptors and behavior, both immediate
and over longer time scales.
6) Develop and use a model (e.g., Punnett squares, diagrams, and simulations) as evidence to
demonstrate why asexual reproduction results in offspring with identical genetic information
and sexual reproduction results in offspring with genetic variation.
7) Develop a model using evidence that explains the process of photosynthesis, cellular respiration,
and anaerobic respiration in the cycling of matter and flow of energy into and out of organisms.
Interactions, Energy, and Dynamics
1) Develop a model to depict the cycling of matter, including carbon and oxygen, and the flow of
energy among biotic and abiotic parts of an ecosystem.
Heredity
1) Evaluate and communicate information that chromosomes contain many distinct genes which
code for the production of proteins, impacting the traits of an individual.
2) Construct an explanation to describe how the impact of changes to genes (i.e., mutations)
located on chromosomes may result in harmful, beneficial, or neutral effects to the structure
and function of the organism.
3) Predict the probability of individual dominant and recessive alleles to be transmitted from each
parent to offspring during sexual reproduction and represent the phenotypic and genotypic
patterns using ratios.
Engineering Design
a solution taking into consideration the criteria, constraints, and relevant scientific principles of
the problem that may limit possible solutions.
- Biotic factor – living factor
- Abiotic factor – nonliving factor
- Carbon Cycle – the path carbon travels on earth
- Oxygen Cycle – the path oxygen travels on earth
- Biogeochemical Cycles - the cycles of life on earth
- Decomposition – to break down
- Fossil Fuels – resources created by once living things over thousands of years
- Producer – plants, autotrophs
Photosynthesis – how plants create “make” their own food
using sunlight
Cellular Respiration – how plants and animals “break”
down food for energy(ATP), using oxygen.
Anaerobic Respiration – occurs WITHOUT oxygen
Aerobic Respiration – occurs WITH oxygen
Autotroph – organism that makes its own food in
chloroplasts...plants
Heterotroph – organism that hunts food for energy...animals
Chloroplast - site of photosynthesis occurs in plant cells.
Mitochondria – organelle where cellular respiration occurs
in plant and animal cells.
Chemical Energy – potential energy stored in atom bonds
Mechanical Energy – total energy due to motion
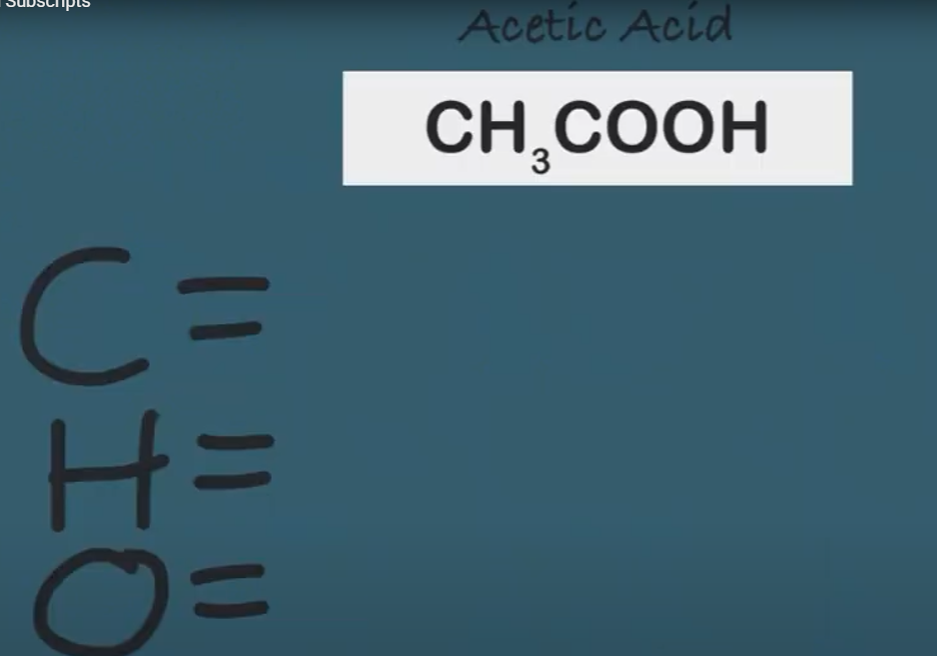
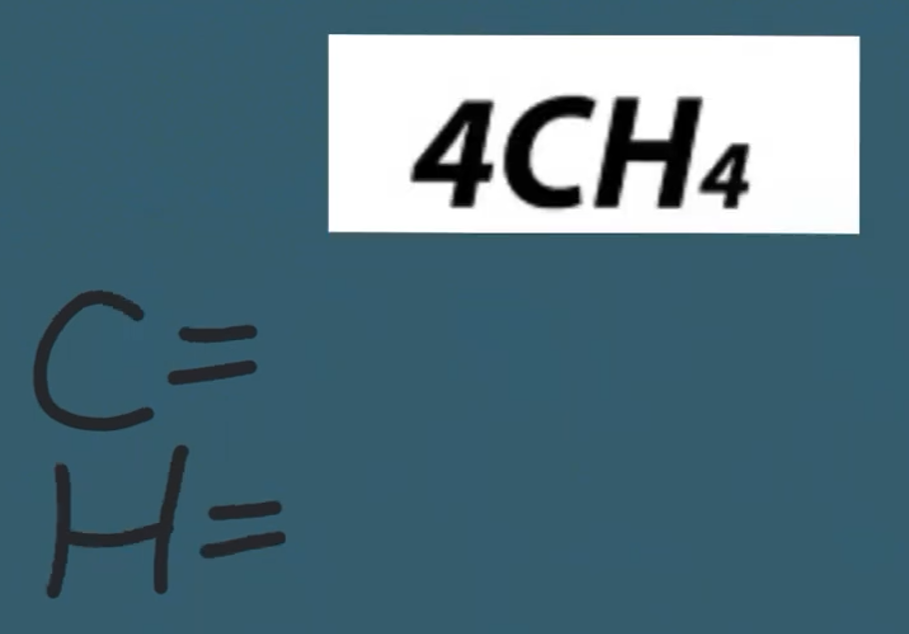
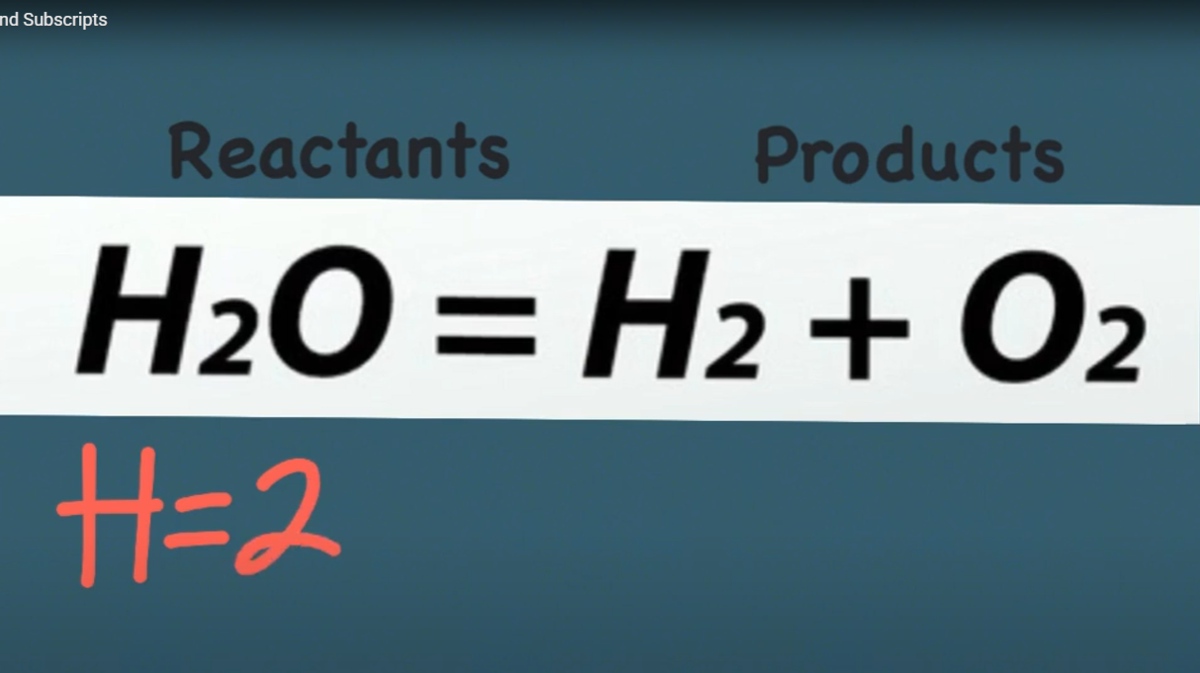
-
Reactant – The atoms that react...to the left
-
Product – The atoms that are produced...to the right
-
Coefficient – The whole # multiplier...BIG #
-
Subscript - How many atoms you have...small #
-
Mass – amount of matter in a given space
-
Chemical Equation - symbolic, mathematical representations of chemical reactions
-
Balanced - the reactants mass and atom count are EQUAL to the products mass and atom count
-
Chemical Reaction – chemical process where reactants interact to form new products or substances...PURE
-
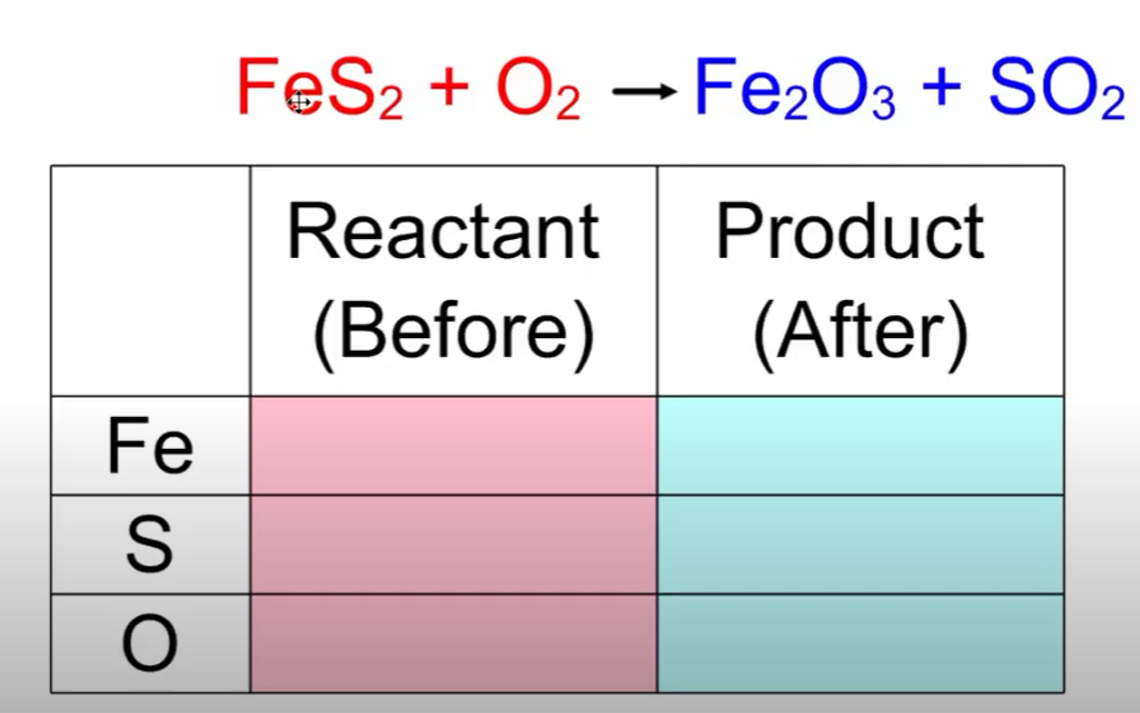
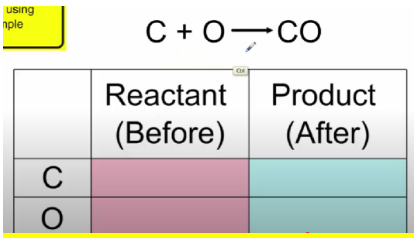
Law of Conservation of Mass – matter cannot be created or destroyed (LOCOM)
- Input - what goes "into" the chemical reaction
- Output - what comes "out" of the chemical reaction
- Temperature – measure of hotness or coldness
- Pressure – physical force exerted on an object
- Intermolecular Attraction – force that determines the physical properties of a substance
- Thermal Energy – energy contained in a system that determines temperature...HEAT
- Triple Point Diagram – diagram that shows the three states of matter for a substance
- Kinetic Energy – the energy of particles in motion
- Open System – can exchange matter and energy with its surroundings..."open"
- Closed System – cannot exchange matter and energy with its surroundings..."sealed"
- Solid – Definite shape, Definite volume, least energy
- Liquid – NO definite shape, Definite volume, mid energy
- Gas – NO Definite shape or volume, most energy
- Release – to give off or go out
- Absorb – to take in
- Solute – dissolved into solvent...salt (less)
- Solvent – dissolves other substances...water (more)
- Chemical reaction – changing substances to totally new substances...A + B = C
- Thermal Energy – hot or cold
- Endothermic – heat absorbed = cooling
- Exothermic – heat released = warming
- Reactants – factors/molecules on the left side
- Products – factors/molecules on the right side
Blooket #1 - https://play.blooket.com/play?hwId=6980cac49dcf52d5ecc20903
- Atom – made of protons, neutrons, and electrons
- Matter – anything that has mass and takes up space
- Element – pure substance made up of only one type of atom
- Molecule – group of atoms chemically bonded together
- Monatomic – made of only one atom and no bonding
- Diatomic – made up of two atoms bonded
- Elemental Molecule – two or more of the same atoms chemically bonded together
- Compound Molecule – two or more different types of atoms chemically bonded together
- Ionic Bond – bond where electrons are transferred
- Covalent Bond – bond where electrons are shared
- Atom – makes up all matter
- Electron – subatomic particle with a nEgative(-) charge
- Proton – subatomic particle with a positive(+) charge
- Neutron – subatomic particle with NO(none) charge
- Atomic Nucleus – made up of protons and neutrons
- Orbital Shell – rings around the nucleus that hold electrons
- Valence Electron – electrons in the outermost shell
- Periodic Table – visual arrangement of chemical elements
- Atomic Number – number of electrons in the element
- Subatomic Particle – smaller than the atom
- Matter – any substance with mass that takes up space
- Property – characteristic that can be observed or measured
- Adaptation- changing to survive in your environment
- Environmental Factor- any factor, whether abiotic or biotic, that influences living organisms
- Inherited Factor- alleles/genes passed down from parent to offspring
- Growth- process of increasing in size and maturity
- Population Density – number of organisms in a given area
- Carry Capacity – number of organisms an area can healthily support
- Stimulus- something that causes a response
- Nutrients- essential substances organisms need
- Natural Selection- only the fittest and strongest survive
- Resistant- unable to be affected by
- Modification- a change made to a system
Class Codes
4th - YC5K2
5th - 586EJ
6th - RV7AH
7th - P2KM9
- Mutation – change in DNA that creates new PROTEIN
- Gene – section of DNA that codes for protein
- DNA – found in the nucleus of each cell
- Protein – molecule that gives you your trait
- Helpful Mutation – beneficial..change that makes you stronger
- Harmful Mutation – hurts..change that harms you
- Neutral Mutation – harmless..change that doesn't affect you
- Insertion Mutation – genes added to your DNA
- Deletion Mutation – genes removed from your DNA
- Substitution Mutation – genes replaced in your DNA
- Advantage – a good circumstance
- Disadvantage - a bad circumstance
- Genetic Variation – organisms with different genes
- Reproduction – process of a creating offspring
- Asexual Reproduction – reproducing with one parent cell
- Sexual Reproduction – reproducing with two parent cells
- Offspring – the product of reproduction
- Survival – continuing to live or exist
- Genotype – alleles you inherit from parents
- Heredity – DNA you received from your parents
- Chromosome – made up of condensed DNA
- Gene – portion of DNA
- DNA – genetic material inherited from parents
- Dominant – stronger gene, allele
- Recessive – weaker gene, allele
- Trait – an inherited characteristic, expressed
- Protein – made by genes, produces traits
- Pairs – set of 2 chromosomes
- Hierarchy – organized from simple to complex
Amazon.com: Cell Fie Funny Cellfie Selfie Biology Science Teacher T-Shirt : Clothing, Shoes & Jewelry
- Clip #1 - Bing Videos (Blue Fugates) Mystery of the Fugate Family: The Blue People of Kentucky - YouTube (youtube-nocookie.com)
- Clip #2 – Hemingway Cats Why So Many Six-Toed Cats Live At The Hemingway House - YouTube
- Clip #3 – Moken People How Moken children see with amazing clarity underwater - Inside the Human Body - BBC One - YouTube
- Gene- specific sequence of DNA...codes for a trait
- Trait- characteristic passed from one generation to another
- Allele- different versions of the trait (eye colors)
- Dominant trait- stronger gene/allele (capital letters)
- Recessive trait- weaker gene/allele (lower case letters)
- Phenotype- physical trait, what you look like (blue eyes)
- Genotype- letter set inherited (BB, Bb, bb)
- Probability- Odds...1/4, 2/4, 3/4, 4/4
- Heterozygous- different genes or letters (Bb, Tt, Rr)
- Homozygous- same genes or letters (TT, RR, EE)
- Punnett square- mathematical model for inheritance
- Stimulus – something that causes an organism to react
-
- Taste – using taste buds to find safe food
-
- Smell – using olfactory senses to signal the brain
-
- Sight – using the eyes to stay safe in an environment
-
- Mechanoreceptor – cell detecting pain, pressure, and sound
-
- Thermoreceptor – cell detecting heat and cold (thermal energy)
-
- Chemoreceptor – cell detecting scent and taste
-
- Photoreceptor – cell detecting "light"
-
- Nervous System – the brain, spinal cord, and nerves
-
- Neuron – a nerve cell
- Body system- made up of specialized organs that perform a specific function/job
-
- Sensation- the ability to feel and sense one's environment
-
- Locomotion- the ability to move
-
- Interdependence- to work together...systems always work together
-
- Homeostasis- a state of balance among all body systems
-
- Equilibrium- a state of balance (Ex. 10 = 10)
-
- Nutrients – food carried by the blood to your cells
-
- Immunity – not able to contract or be infected by
-
- Carbon Dioxide – waste gas exhaled by humans
- Blood – tissue that carries oxygen, carbon dioxide, and nutrients to all cells
Turn to a New page in your journal:
- Cell- basic unit of life
- Tissue- group of similar cells
- Organ- group of tissues working together
- Organ system- 2 or more organs working together
- Organism- made of multiple organ systems working together
- MULTI-CELLULAR- made of 2 or more cells
- UNI-CELLULAR- made of only 1 cell
- Specialized Cell- cell doing a specific job
- Independent – functions alone
- Interdependent – depends on others
https://www.gimkit.com/join/68b970cdff123d1fca80d399
-
Autotroph – "auto"matically makes own food (chloroplast)
-
Heterotroph – "hunter" consumer (no chloroplast)
-
Asexual Reproduction – reproduction with ONE parent cell
-
Sexual Reproduction – reproduction with TWO parent cells
-
Prokaryote – No nucleus (NO!)
-
Eukaryote – Has a nucleus (You DO!)
-
Terrestrial – lives on land (TREE)
-
Aquatic – lives in water (Aqua=water)
-
Unicellular – only one cell
-
Multicellular – 2 or more cells
- Cell Membrane- "Semi-permeable" layer surrounding all cells
-
- Cell Wall- "Rigid" covers PLANT Cells
-
- Cytoplasm- "Jelly-like" fluid and the organelles.
-
- Mitochondria- "Powerhouse" of the cell...ENERGY producer!
-
- Nucleus- DNA found here; "brain/control center" of the cell
-
- Ribosomes- Make "protein, protein, protein"
-
- Endoplasmic Reticulum- "Passageways" to transport proteins
-
- Golgi Bodies- packages proteins..."packager"
-
- Chloroplasts- Green, PLANT cells, make food..."photosynthesis!"
-
- Large Vacuoles- Store "water" for the PLANT cell..."vacupool"
-
- Lysosomes- The "cleaner" of the cell..."Lysol"

